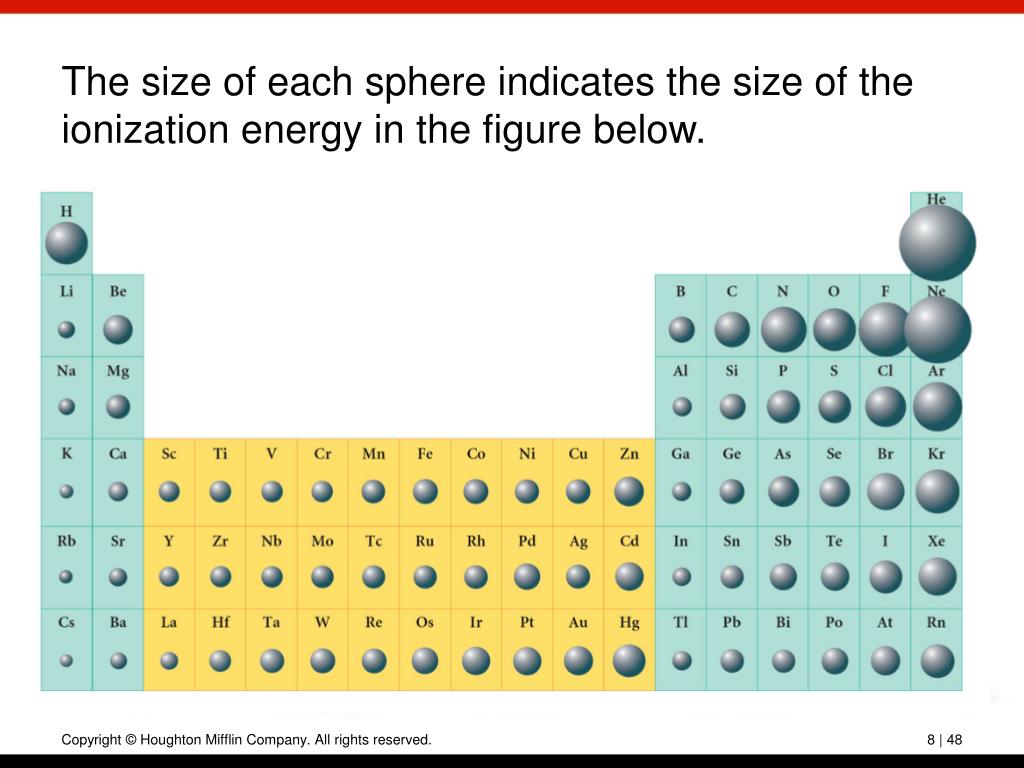
The next element down, magnesium, is expected to have exactly the same arrangement of electrons in the n = 3 principal shell: s 2. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n = 2.ī Thus beryllium has an s 2 electron configuration. Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum number of its valence shell, its valence orbitals, and the number of valence electrons in each orbital as superscripts.Ī The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons.Locate the nearest noble gas preceding each element and identify the principal quantum number of the valence shell of each element. Identify the block in the periodic table to which the group 2 elements belong.
Use the periodic table to predict the valence electron configuration of all the elements of group 2 (beryllium, magnesium, calcium, strontium, barium, and radium).Īsked for: valence electron configurations For elements after No, the electron configurations are tentative. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.\( \newcommand\): Electron Configurations of the Elements. (See the above or below periodic table to see the valence electrons of inner transition metals). Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The inner transition elements have incomplete f-subshells and they are very close to the outer s-subshell. In an electron configuration under the general form of ns1, the n represents the period number and the 1 represents the group number, and therefore, the element would be in group 1. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. ns2, np3 means valence electrons 5 group 15 in the periodic table contains valance shell electrons 5 Nitrogen 7 1s2, 2s2, 2p3 P 13 1s2, 2s2, 2p6. So, the electrons of both d-subshell and s-subshell behave like valence electrons. As we go down the periodic table, the valence shell gets larger. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. The presence of eight electrons in the valence shell of an atom imparts stability to that particular atom. Valence electrons or outer-shell electrons are indicative of an atom’s reactivity. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. The electronic configuration of nitrogen is \(2,\,8,\,7.\) Chlorine has \(7\) electrons present in its valence shell. Preceeding the noble gases are two chemically important groups of non-metals such as halogens (Group 17) and the chalco gens (Group 16). This configuration is highly stable and hence noble gases exhibit very low chemical reactivity. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? At the end of each period is a noble gas element with a closed valence shell ns2 np6 configuration. Hence, magnesium has 2 valence electrons.
Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). The magnesium element has 2 electrons in outermost orbit.
